
Does sugar cause inflammation? Not directly.
But the connection between sugar and inflammation is real—just not in the way most people think.
Here’s what’s actually happening in your body, why the confusion exists, and what this means if you’re trying to reverse prediabetes.
Does Sugar Cause Inflammation? The Short Answer
Sugar doesn’t directly cause inflammation. What sugar does is cause insulin resistance. And insulin resistance creates a metabolic environment where inflammation can develop—but it’s not a direct cause-and-effect relationship.
The distinction matters because if you’re trying to reverse prediabetes, understanding the actual mechanism helps you address the real problem instead of chasing symptoms.
What Research Actually Shows About Sugar and Inflammation
When scientists look at does sugar cause inflammation, the evidence is mixed and limited. A 2018 meta-analysis by Della Corte and colleagues examined all available intervention studies and found that “limited evidence does not support the hypothesis that dietary sugar contributes to subclinical inflammation.”
But that doesn’t mean sugar is harmless.
What the research consistently shows is that high sugar intake causes insulin resistance. And insulin resistance—the condition where your cells stop responding properly to insulin—creates metabolic dysfunction that involves inflammatory processes.
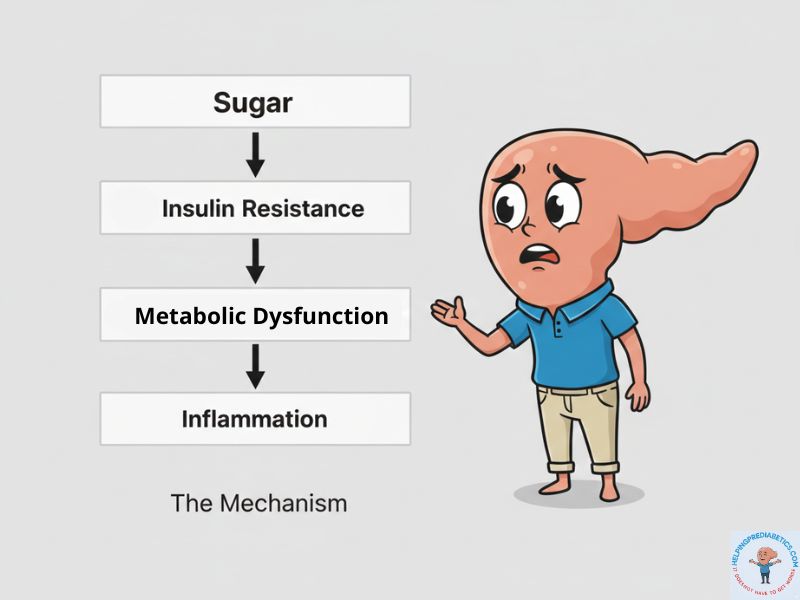
It’s not: Sugar → Inflammation
It’s: Sugar → Insulin Resistance → Metabolic Dysfunction (which involves inflammation among other things)
The pathway is more complex than direct causation.
Why Sugar Source Matters for Inflammation

Here’s where it gets more nuanced: not all sugar sources affect your body the same way.
Fructose from whole fruit? Research shows it can actually reduce inflammation markers. The fiber, nutrients, and food matrix matter.
Fructose from soda and fruit juice? That’s associated with increased inflammation markers. Same sugar molecule, different metabolic effect because of how it’s delivered. This is why answering does sugar cause inflammation requires nuance about source and delivery method.
And let’s be clear: orange juice is not fruit. It’s fruit stripped of its fiber. Without that fiber, the sugar enters your bloodstream at nearly soda-like speed. This is why studies consistently link juice to higher inflammation markers and insulin resistance, while whole oranges are neutral or protective. The fruit matters less than what you’ve done to it.
When people ask “does sugar cause inflammation,” they’re usually thinking about processed sugar in sodas, desserts, and packaged foods. And there, the evidence is stronger—but it’s still working through insulin resistance as the primary mechanism, not direct inflammation.
Why Is Sugar Bad: The Truth Behind the Confusion
So if the answer to does sugar cause inflammation is ‘not directly,’ why are there talks about sugar causing inflammation?
Part of the confusion comes from conflating correlation with causation. People who eat high-sugar diets often have elevated inflammation markers. But they also often have obesity, sedentary lifestyles, and poor overall diet quality—all of which independently contribute to inflammation.
Separating sugar’s specific effect from these other factors is difficult.
The other part comes from decades of misleading information about sugar—first promoting it as harmless, then demonizing it without nuance.
History of Sugar Industry: The 1950s Boom and What Came After

In the 1950s, the sugar industry launched massive marketing campaigns promoting sugar as pure energy. Harmless. Natural. Necessary for active living.
Behind the scenes, the industry was doing something else: funding research designed to shift blame away from sugar and toward dietary fat as the cause of heart disease. Internal documents analyzed by Kearns and colleagues in a 2016 JAMA Internal Medicine study revealed how the Sugar Research Foundation sponsored research reviews that downplayed sugar’s role in coronary heart disease while emphasizing saturated fat.
This wasn’t accidental. The sugar industry actively worked to shape scientific consensus in their favor.
By the 1970s, the Federal Trade Commission began cracking down on misleading sugar advertising. The narrative that “sugar is just empty calories—harmless if you don’t overdo it” started to collapse as research accumulated showing metabolic consequences beyond simple calorie excess.
But when that original narrative fell apart, the pendulum swung hard in the opposite direction. Now sugar gets blamed for everything—often without the nuance that actual research requires.
The truth sits somewhere between “sugar is harmless” and “sugar directly causes all metabolic disease.” Understanding where it actually sits helps you address the real mechanisms.
Sugar and Inflammation: The Real Mechanism Through Insulin Resistance
Here’s what actually happens when you consume high amounts of sugar repeatedly over time:
Your blood glucose rises. Your pancreas releases insulin. Insulin signals your cells to absorb glucose from your bloodstream.
When this happens occasionally, your body handles it fine.
When this happens multiple times a day, every day, for years—several metabolic changes occur simultaneously:
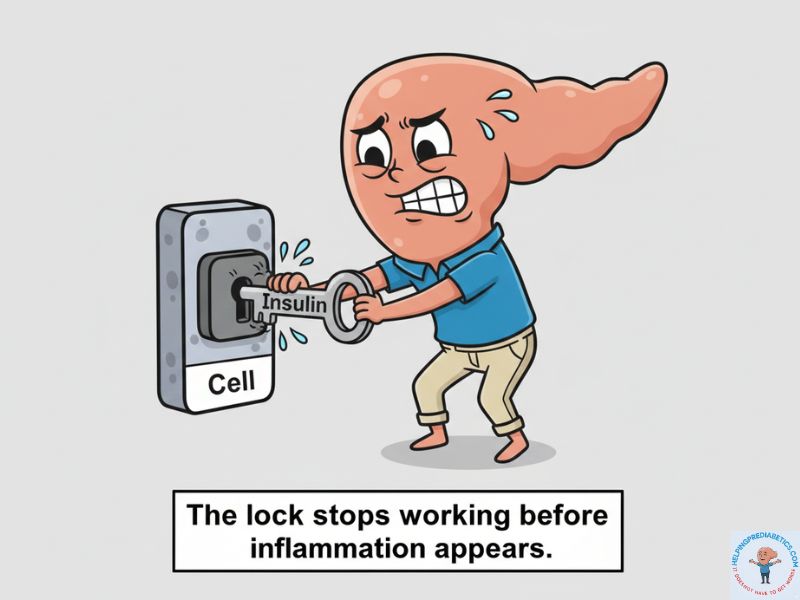
Think of insulin as a key turning a lock to let glucose into your cells. When that key turns occasionally, the lock works perfectly. But when too much energy floods in—especially from fructose being converted to fat in your liver—tiny fat droplets start accumulating inside the cell itself. Those droplets jam the lock mechanism.
The key still turns. But the door doesn’t open as easily anymore. Your pancreas has to produce more keys (more insulin) just to get the same amount of glucose inside. This is insulin resistance at the cellular level.
And here’s what compounds the problem:
Chronic elevated insulin levels. Not just spikes after meals, but baseline insulin that stays elevated even when fasting. This chronic hyperinsulinemia is both a marker and a driver of insulin resistance.
Ectopic fat accumulation. When your liver and muscles are repeatedly flooded with glucose and can’t store it all as glycogen, excess energy gets converted to fat and stored in places it doesn’t belong—inside liver cells (hepatic steatosis) and muscle cells (intramyocellular lipids). This ectopic fat directly interferes with insulin signaling—those fat droplets jamming the lock.
Cellular adaptation. Your cells aren’t failing. They’re adapting to a message they hear too often. When insulin signals arrive constantly, cells turn down the volume. It’s not a defect—it’s your body protecting itself from chronic overstimulation.
Visceral fat expansion. Excess energy intake (particularly from sugar and refined carbohydrates) gets stored as visceral fat—the metabolically active fat around your organs. This fat tissue secretes inflammatory molecules (adipokines) that further worsen insulin resistance, creating a self-reinforcing cycle.
All of these processes work together. Insulin resistance isn’t just about meal-by-meal glucose spikes—it’s about sustained metabolic dysfunction involving baseline insulin levels, fat storage patterns, and tissue-level changes in how your body processes energy.
Your body is already showing you these signals:
The energy dips after lunch, the hunger that returns an hour after eating, the brain fog mid-afternoon. These aren’t character flaws. They’re your body mirroring back what your metabolism needs—fewer insulin demands, more recovery time between meals, a different frequency pattern.
This is why insulin resistance is the core metabolic dysfunction in prediabetes. It’s what causes:
- Elevated fasting blood glucose
- Higher HbA1c levels
- Increased cardiovascular disease risk
- Difficulty losing weight
- Energy crashes and cravings
And yes, insulin resistance does involve inflammatory pathways. But inflammation is part of the metabolic dysfunction, not the starting point.
What This Means For Reversing Prediabetes
If you’re prediabetic, understanding this mechanism matters.
You’re not trying to “reduce inflammation” as a primary goal. You’re trying to reverse insulin resistance—which addresses the root metabolic dysfunction.
That happens through:
Reducing the frequency and magnitude of insulin demand. Not just the amount of sugar, but how often your body has to produce large amounts of insulin throughout the day. This is about reducing your body’s recovery demand—how often it has to respond, how much time it gets to return to baseline between insulin signals. Meal timing, carbohydrate type and quantity, and eating patterns all matter.
Reducing ectopic fat stores. Through sustained caloric balance, physical activity, and giving your liver and muscles time to clear stored fat. This restores insulin signaling at the cellular level—clearing out those fat droplets jamming the lock.
Restoring your cells’ insulin sensitivity. Through consistent eating patterns, movement that depletes muscle glycogen, sleep that allows metabolic recovery, and stress management that doesn’t trigger constant cortisol-driven glucose release. Your cells aren’t broken—they’re adapting. You’re helping them adapt back.
Addressing visceral adiposity. Not through targeted fat loss (which isn’t possible), but through overall metabolic improvement that naturally reduces visceral fat as insulin resistance improves.
Here’s what’s important about “anti-inflammatory” foods:
Many of the foods often recommended for their anti-inflammatory properties—vegetables high in fiber and polyphenols, omega-3 fatty acids from fish, berries, leafy greens—also happen to improve insulin sensitivity. The mechanisms overlap significantly.
Adding these foods is wonderful for your overall health. But if your cells are still being flooded with glucose and insulin six times a day, those foods alone can’t undo the insulin resistance pattern. The priority is reducing the signal frequency—how often your body has to mount an insulin response. Then the beneficial foods have metabolic space to do their work.
It’s not that anti-inflammatory approaches are wrong. It’s that they work best when the underlying frequency problem is also being addressed. You need both: beneficial foods AND pattern changes that reduce insulin demand and allow your body to clear stored metabolic dysfunction.
Why Side Effects of Sugar Go Beyond Simple Calories

When people search for “sugar side effects” or “why is sugar bad for you,” they’re often trying to understand why sugar feels different from other foods—why it seems to trigger cravings, energy crashes, and weight gain in ways that other calorie sources don’t.
The insulin resistance mechanism explains this.
Sugar (particularly in liquid form like sodas, or in processed foods where it’s combined with refined flour and fat) creates rapid glucose spikes. These spikes demand large insulin responses. Over time, this pattern creates:
Energy instability. High insulin drives glucose into cells rapidly, often creating reactive hypoglycemia—the crash that follows the spike. This triggers hunger and cravings for more quick energy, perpetuating the cycle.
Leptin resistance. Chronic high insulin interferes with leptin signaling—the hormone that tells your brain you’ve had enough food. This is why high-sugar diets often lead to overeating even when caloric needs are met.
Preference shift. Your taste receptors adapt to high sugar intake, making naturally sweet foods (like fruit) taste less satisfying. You need more sweetness to trigger the same reward response.
Fat storage bias. High insulin is a storage signal. When insulin is chronically elevated, your body preferentially stores energy as fat rather than making it available for use. This is why losing weight with insulin resistance feels nearly impossible—the energy is being locked away in storage rather than made available for cellular use.
These effects aren’t just about calories. They’re about the metabolic environment sugar creates when consumed frequently in processed forms.
Effects of Sugar on the Body: The Full Picture

The effects of sugar on your body depend entirely on context:
Amount matters. Small amounts of sugar in the context of a meal with protein, fat, and fiber create a modest insulin response that your body handles easily. Large amounts of sugar (particularly in liquid form or without other macronutrients) create metabolic stress.
Source matters. Whole fruit delivers fructose with fiber, which slows absorption and provides beneficial nutrients. Soda delivers fructose rapidly with no buffer, creating metabolic burden.
Frequency matters. Occasional sugar consumption allows your body to return to baseline between episodes. Constant grazing on sugary foods and drinks keeps insulin chronically elevated, preventing metabolic recovery.
Individual tolerance matters. Someone who is already insulin resistant will respond differently to the same sugar intake than someone with normal insulin sensitivity. What’s metabolically tolerable for one person may drive dysfunction in another.
This is why blanket statements about sugar—whether “it’s harmless” or “it directly causes disease”—miss the complexity of how metabolism actually works.
The truth about sugar is that in moderate amounts from whole food sources, consumed occasionally by metabolically healthy individuals, it’s well-tolerated. But in large amounts from processed sources, consumed frequently by people who are already insulin resistant, it perpetuates and worsens metabolic dysfunction.
Your goal if you’re prediabetic isn’t to eliminate sugar entirely. It’s to restore insulin sensitivity so your body can handle normal amounts of carbohydrate without dysfunction.
The Bottom Line: Does Sugar Cause Inflammation?

Does sugar cause inflammation? Not directly.
Does sugar cause insulin resistance? Yes. Consistently. When consumed frequently in large amounts, particularly from processed sources.
Does insulin resistance involve inflammatory processes? Yes. As part of broader metabolic dysfunction involving chronic hyperinsulinemia, ectopic fat storage, and cellular adaptation.
The distinction matters because chasing “anti-inflammatory” interventions while continuing patterns that drive insulin resistance won’t fully reverse prediabetes. You have to address the actual mechanism—restoring insulin sensitivity through reduced insulin demand, cleared ectopic fat stores, and metabolic recovery.
Understanding the accurate answer to does sugar cause inflammation helps you address the root cause instead of chasing symptoms.
Sugar isn’t innocent. The 1950s narrative that it was just “empty calories” ignored real metabolic consequences. But it’s also not the direct villain it’s often made out to be in oversimplified “sugar causes inflammation” claims.
Understanding what’s actually happening in your body—the real mechanism involving insulin resistance, not just vague inflammation—gives you the power to address the root cause instead of managing symptoms.
That’s how prediabetes gets reversed. Not through fear of sugar. Not through adding anti-inflammatory foods while keeping patterns unchanged. Through understanding insulin resistance and systematically restoring your body’s ability to handle carbohydrate metabolism properly.
Want to understand exactly how to work with your body to reverse insulin resistance?
Your Body Is Talking: A Pharmacist’s Guide to Stopping Prediabetes is a free guide that explains exactly what’s happening — and what to do about it. No meal plans. No shame. Just clarity.
MEDICAL DISCLAIMER
This article is for educational purposes only and does not constitute medical advice. If you have prediabetes or any metabolic condition, work with your healthcare provider before making dietary changes. Individual responses vary based on genetics, medication use, and other health factors.
REFERENCES
1. Della Corte KW, Perrar I, Penczynski KJ, et al. Effect of Dietary Sugar Intake on Biomarkers of Subclinical Inflammation: A Systematic Review and Meta-Analysis of Intervention Studies. Nutrients. 2018;10(5):606. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5986486/
2. Kearns CE, Schmidt LA, Glantz SA. Sugar Industry and Coronary Heart Disease Research: A Historical Analysis of Internal Industry Documents. JAMA Intern Med. 2016;176(11):1680-1685. Kearns et al., 2016 – Sugar Industry Historical Analysis https://pubmed.ncbi.nlm.nih.gov/27617709/
3. Samuel VT, Shulman GI. The pathogenesis of insulin resistance: integrating signaling pathways and substrate flux. J Clin Invest. 2016;126(1):12-22. https://www.jci.org/articles/view/77812
